Literature searches and Systematic Reviews of published animal studies
Good literature searches are an integral part of planning animal research and testing: please see the section on literature searching in the PREPARE guidelines and the recorded webinars listed here. Some core resources are cited here. The section of the Norecopa website on databases and guidelines will also be helpful.
- Bugasjka et al. (2025): How long does it take to complete and publish a systematic review of animal studies?
- Ineichen et al. (2024): Systematic review and meta-analysis of preclinical studies
- van der Mierden et al. (2024): Laboratory animals search filter for different literature databases: PubMed, Embase, web of Science and PsycINFO
- The EURL ECVAM Search Guide
- How to conduct a literature search by Alice Tillema, Radboud University
- 3Rs search terms (a list compiled by the Kornhauser Health Sciences Library, University of Louisville)
- Advice, concepts and terminology (compiled by AWIC, USDA)
- Searching for information on 3Rs and 3Rs Resources (a presentation by Michelle Hudson-Shore (see below), FRAME (now Replacing Animal Research)
- Enhancing search efficiency by means of a search filter for finding all studies on animal experimentation in PubMed
- A search filter for increasing the retrieval of animal studies in Embase
- SYRCLE's risk of bias tool for animal studies
- PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses), which includes a checklist. Page et al. (2021) describes the updated guideline.
CAMARADES Berlin have published a Preclinical Systematic Review (SR) Wiki, which also contains useful information on how to set up a literature search (regardless of whether or not you plan to conduct an SR).
Systematic reviews of published animal experiments are a useful aid in planning research and can also be used to extract new conclusions from experiments which have already been performed (so-called Synthesis of Evidence), thereby avoiding the need for new ones.
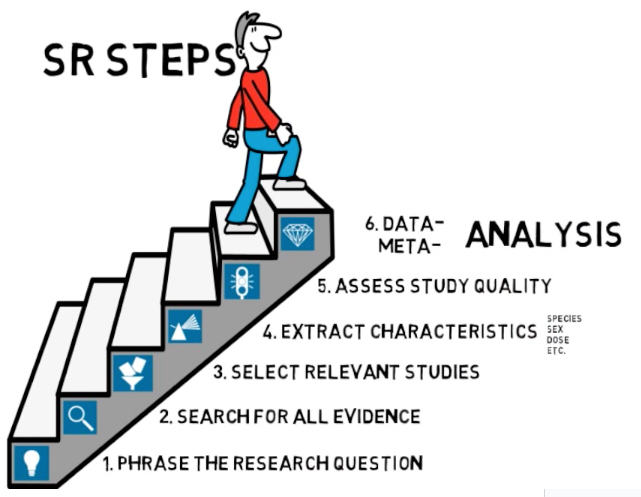
SYRCLE (SYstematic Review Centre for Laboratory animal Experimentation) at Radboud Medical University Center, Nijmegen has produced an excellent video explaining the principles behind systematic reviews. The rationale behind systematic reviews is explained by Professor Merel Ritskes-Hoitinga. Benefits of preclinical systematic reviews (webinar by Merel Ritskes-Hoitinga, 27 October 2020)
Two presentations about systematic reviews from members of SYRCLE at the EUSAAT 2016 congress :
- Systematic Reviews of animal studies equal the implementation of the 3Rs (Merel Ritskes-Hoitinga)
- Improving animal research using a science driven approach: Systematic reviews of animal studies (Judith van Luijk)
Resources about systematic reviews
- Introduction to systematic reviews (NC3Rs webpage)
- Systematic Review Toolbox (SR Toolbox)
- Systematic reviews in animal research: Launch of the CAMARADES-NC3Rs Systematic Review Facility (SyRF)
- The Evidence-Based Research Network
- Evidence Synthesis International
- VetSRev - a database of veterinary systematic reviews
- BestBETs for Vets - CAT database of clinical questions
- Peer review training and guides (EQUATOR network website)
- CAMARADES Zurich
Examples of Systematic Reviews of relevance to animal research
- Wolter et al. (2021): Systematic review on the reporting accuracy of experimental details in publications using mouse femoral fracture models
- Franco et al. (2019): Methodological standards, quality of reporting and regulatory compliance in animal research on amyotrophic lateral sclerosis: a systematic review
- Franco et al. (2012): Animal Welfare in Studies on Murine Tuberculosis: Assessing Progress over a 12-Year Period and the Need for Further Improvement
- Franco et al. (2012): "How Sick Must Your Mouse Be?”—An Analysis of the Use of Animal Models in Huntington's Disease Research
Other references
- Heron L, Buitrago-Garcia D, Ipekci AM et al. (2023): How to update a living systematic review and keep it alive during a pandemic: a practical guide. Syst Rev 12, 156.
- Hunniford VT, Montroy J, Fergusson DA, Avey MT, Wever KE, McCann SK et al. (2021) Epidemiology and reporting characteristics of preclinical systematic reviews. PLoS Biol 19(5): e3001177.
- Karunananthan et al., (2020): When and how to replicate systematic reviews
- Hollier (2020): Research Basics: Using Boolean Operators to Build a Search
- van der Naald M et al. (2020): Publication rate in preclinical research: a plea for preregistration
- Brock (2019): How researchers can improve the quality of systematic reviews
- Cramond et al. (2019): The development and evaluation of an online application to assist in the extraction of data from graphs for use in systematic reviews
- van der Mierden et al. (2019): Software tools for literature screening in systematic reviews in biomedical research
- Sena ES et al. (2014): Systematic reviews and meta-analysis of preclinical studies: why perform them and how to appraise them critically
- Korevaar, Hooft & ter Riet (2011): Systematic Reviews and meta-analyses of preclinical studies: publication bias in laboratory animal experiments.
- Würbel (2007): Publications should include an animal-welfare section
The University of Bergen's Vivarium and Norecopa arranged a workshop in Voss, Norway on 27-28 May 2015 entitled Systematic Reviews and Harm-Benefit Assessment of Animal Experiments. The workshop gave theoretical and practical training in how to conduct literature searches and systematic reviews of the laboratory animal science literature. In addition, it gave an introduction to the harm-benefit assessment of animal experiments. Documentation of literature searches and harm-benefit assessment are two requirements of EU's Directive 2010/63. The theoretical and practical training in systematic reviews were led by representatives of SYRCLE. The discussion of harm-benefit assessment was led by Dr. Penny Hawkins, Head of the Research Animals Department at the RSPCA, UK and Dr. Aurora Brønstad, University of Bergen, who was the co-chair of an AALAS-FELASA working group on this subject. Advice on literature searching and available databases was provided by Adrian Smith. Several of these resources have been produced by Norecopa. The workshop took place at Fleischer's Hotel, Voss (more information about the workshop). Participants brought their own personal computers with the program Review Manager 5 from the Cochrane Collaboration installed.
The organisers gratefully acknowledge financial support for this workshop from the Research Council of Norway.
Searching for information on 3Rs and 3Rs Resources is the title of a presentation that was included in the FRAME/VetBioNet Training School in Experimental Design and Statistical Analysis of Bioscience and Biomedical Experiments, held on 9-11 January 2019 at the University of Nottingham, U.K. It aims to give scientists an awareness of the need to conduct effective searches and the problems associated with searching, as well as basic search skills and increased knowledge of available resources. Norecopa has been given permission to publish the presentation here. A slightly older version is available on YouTube.
The author, Dr. Michelle Hudson-Shore, is dedicated to improving training provision and contributing to learning and development within organisations and institutions through her work with colleagues at the Centre for Applied Bioethics (CAB), University of Nottingham. CAB is very interested in developing and providing training in 3Rs, ethics, implementation of animal experimentation legislation and the social impact of research, both for animal related studies and scientific research more broadly. If you feel that you and your research group could benefit from professional development in these areas please get in touch with Dr Michelle Hudson-Shore or Dr Kate Millar.
Thanks for your feedback! Please note that we cannot reply to you unless you send us an email.
What are you looking for?
We value your feedback so we can improve the information on the page. Please add your email address if you would like a reply. Thank you in advance for your help.!
Please contact us by email if you have any questions.
