Research on wildlife and wild fish
Field researchers in Norway should also read the Norwegian version of this page.
The sections on fish, cephalopods and other aquatic animals may also be relevant.
Research on wild-living animals places special demands on experimental design, since it takes place under uncontrollable environmental conditions. There are also a range of ethical problems in addition to those which apply to all animal research, since it is often difficult to monitor the welfare of animals after their release.
Norecopa har arranged two international consensus meetings about field research:
- May 2008: Harmonisation of the Care and Use of Animals in Wildlife Research
All the presentations from the meeting, as well as a consensus document written by the participants, can be downloaded. The document describes the status and challenges related to wildlife research. At the meeting, Dr. Penny Hawkins presented an overview of guidelines for wildlife research. - October 2017: Harmonisation of the Care and Use of Wild and Domestic Mammals and Birds in Field Research
Norecopa has a collection of presentations on wildlife research here.
Guidelines and other resources
- Soulsbury et al. (2020): The welfare and ethics of research involving wild animals: A primer.
- Norecopa's database 3R Guide contains a list of guidelines for wildlife research.
- Guidance on planning experiments is available in the PREPARE guidelines.
- Advice on wildlife research from the NC3Rs
- Guidance documents and regulatory advice from ASRU (Animals in Science Regulation Unit, UK). This includes
- Guidelines on the care and use of wildlife from the CCAC (March 2023) - and a Swedish-language summary produced by the Swedish 3Rs Center
- Courses for wildlife researchers
- Dr. Miriam Zemanova of the University of Technology, Sydney, has created a website on 3Rs Principles in Wildlife Research. It contains examples of peer-reviewed studies that implemented non-lethal or non-invasive methods and that can be used as guidance. The site also describes the importance of wildlife research, and potential animal welfare issues. The website includes a useful graphic, summarising ways in which the three Rs can be implemented.
See also: Beyond the lab: Animal welfare and 3Rs in wildlife research, UFAW conference, (Miriam Zemanova), 28 June 2022 (2:50:30 into the recording) - Model Wildlife Protocol developed by the Ornithological Council and the American Society of Mammalogists
- The STRANGE guidelines may be used to identify, and mitigate, potential sampling biases in field research.
- Guidelines for the Care and Use of Insects in Research from the Insect Welfare Research Society
- Norecopa's newsletter no. 4-2022 had a section entitled Focus on wildlife resources
- Is wildlife research "second-rate science"? What can lab animal and field scientists learn from one another? A 16-minute presentation by Adrian Smith, held at the 11th World Congress on Alternatives and Animal Use, August 2021.
- AviSample Network (database of bird sample descriptions from all over the world)
- European Observatory of Wildlife (EOW)
- Wildlife Pathology Unit at the Royal (Dick) School of Veterinary Studies, Edinburgh
Other literature references
- A collection of papers on wildlife diseases and research from Acta Veterinaria Scandinavica
- Browning et al. (preprint): Assessing the suitability of welfare indicators for wild animals
- Kreeger, Arnemo, Caulkett, Hampton & Meyer (2023): Handbook of Wildlife Chemical Immobilization
- Special issue of Biology on "Anaesthetics and Analgesics Used in Aquatic Species" (2023)
- Zemanova MA (2020): Towards more compassionate wildlife research through the 3Rs principles: moving from invasive to non-invasive methods
- Matthew Witt et al. Refining the tagging of wild fish and sharks (project description 2021-2024).
- Sharma et al. (2020): Conservation and people: Towards an ethical code of conduct for the use of camera traps in wildlife research
- Colloff et al. (2024): Use of an expert elicitation methodology to compare the welfare aspects of two approaches for blood sampling European badgers (Meles meles) in the field.
- Special issue of the journal Animals (2020) entitled Mammal Trapping, Wildlife Conservation, and Animal Welfare
- Zemanova MA (2019): Poor implementation of non-invasive sampling in wildlife genetics studies
- The ARROW guidelines (Animals in Research: Reporting on Wildlife) (Field et al., 2019)
- Chinnadurai et al. (2016): Best-Practice Guidelines for Field-Based Surgery and Anesthesia of Free-Ranging Wildlife. I. Anesthesia and Analgesia
- Chinnadurai et al. (2016): Best-Practice Guidelines for Field-Based Surgery and Anesthesia of Free-Ranging Wildlife. II. Surgery
- Advice Note on working with animals taken from the wild. Home Office UK, July 2016. This document also covers situations where wild animals are captured for identification purposes and then set free again.
- UK Home Office checklist for applications for working with wild animals (August 2016)
- Falendysz et al. (2021): Outside the box: working with wildlife in biocontainment
- Lindsjö J et al. (2019): The dividing line between wildlife research and management - implications for animal welfare. Frontiers in Veterinary Science.
- Arnemo JM et al. (2018): Long-Term Safety of Intraperiteonal Radio Transmitter Implants in Brown Bears (Ursus arctos). Frontiers in Veterinary Science.
- Evans AL et al. (2016): Physiological reactions to capture in hibernating brown bears. Conservation Physiology, Volume 4, Issue 1.
- Lindsjö J, Fahlman Å & Törnqvist E (2016): Animal welfare from mouse to moose - implementing the principles of the 3Rs in wildlife research. Journal of Wildlife Diseases, 52(2) Supplement 2016, S65-S77.
- Pacquet PC & Darimont CT (2010): Wildlife conservation and animal welfare: two sides of the same coin? Animal Welfare, 19, 177-190. (Norecopa's consensus meeting is cited twice in this paper).
- Risk assessment concerning marking of wild birds. A report in Norwegian (2024) from the Norwegian Society for Food Safety (VKM).
- Spotswood EN et al., (2012): How safe is mist netting? Evaluating the risk of injury and mortality to birds. Methods in Ecology and Evolution, 3, 29-38.
- Cox RJ et al. (2019): Research with Agricultural Animals and Wildlife. ILAR Journal. doi.org/10.1093/ilar/ilz006
- The Cornell Lab of Ornithology
- A Ten-Stage Protocol for Assessing the Welfare of Individual Non-Captive Wild Animals: Free-Roaming Horses (Equus Ferus Caballus) as an Example
- Animal models - theme issues from the ILAR journal
Fish in research and telemetry tagging methods
Senior Research Scientist Dr. Niels Jepsen at DTU Aqua (the National Institute of Aquatic Resources at the Technical University of Denmark) has written an article entitled Fish in research and telemetry tagging methods for Norecopa and has made available his presentation from a workshop in Dublin in 2015 entitled Capture, Handling and Tagging of Fish for Telemetry Studies. A collection of references to his publications may be found below. More references may be found in the section on marking and identification of fish. A separate section covers resources about anaesthesia and analgesia in fish.
Literature references about tagging fish
- Jepsen, N. & Aarestrup, K. (1999): A comparison of the growth of radio-tagged and dye-marked pike. Journal of Fish Biology 55, 880-883.
- Jepsen, N, Davis, L.E., Schreck, C.B. & Siddens, B. (2001): The physiological response of chinook salmon smolts to two methods of radio-tagging. Transactions of the American Fisheries Society 130, 495-500.
- Jepsen, N., A. Koed, E. B. Thorstad & E. Baras (2002): Surgical implanting of telemetry transmitters in fish. How much have we learned? Hydrobiologia.483, 239-248.
- Jepsen, N. (2003): Long-term retention of radiotransmitters in pikeperch (Stizostedion lucioperca). Journal of Fish Biology 63, 260-262.
- Jepsen, N., Schreck, C., Clement, S. & Thorstad, E. (2004): A brief discussion of the 2% tag/bodymass rule. Aquatic telemetry: advances and applications. M. T. Spedicato, G. Marmulla, G. Lembo (eds.). FAO – COISPA, Rome. Pp 255-259.
- Jepsen, N., Mikkelsen, J. S. & Koed, A. (2008): Effects of tag and suture type on survival and growth of brown trout with surgically implanted telemetry tags in the wild. Journal of Fish Biology 72 , 594-602.
- Jepsen, N., Christoffersen, M. & Munksgaard, T. (2008): The level of predation used as indicator of tagging/handling effects. Fisheries Management and Ecology 15, 365-368.
- Jepsen, N., Boutrup, T.S., Midwood, J. & Koed, A. (2013): Does the level of asepsis impact the success of surgically implanting tags in Atlantic salmon? Fisheries Research 147, 344-348.
- Jepsen, N., Aarestrup, K., Cooke, S.J. (2014): Tagging fish in the field: ethical and procedural considerations. A comment to the recent paper of D. Mulcahy: “Legal, ethical and procedural bases for the use of aseptic techniques to implant electronic devices”, Journal of Fish and Wildlife Management 4, 211-219.
- Jepsen, N., Boutrup, T.S., Midwood, J. & Koed, A. (2014): Fish surgery – a dirty business? Comments to a letter submitted by D. Mulcahy and C.A. Harms. Fisheries Research 156, 6-8.
- Jepsen, N., Thorstad, E.B., Havn, T. & Lucas, M. (2015): The use of external electronic tags on fish – an evaluation of tagging effects. Animal Biotelemetry. 3:49 DOI 10.1186/s40317-015-0086-z
- Cottrill, R.A., Okland, F., Aarestrup, K., Jepsen, N., Koed, A., Hunter, K.J., Butterworth, K.G. & McKinley, R.S. (2006): Evaluation of three telemetry transmitter attachment methods for female silver-phase American eels (Anguilla rostrata Lesueur). Journal of Great Lakes Research 32, 502-511.
- Wargo Rub, A.M., Jepsen, N., Liedtke, T.L., Moser, M. L. & Weber, E. P. S. (2014): Surgical insertion of transmitters and telemetry methods in fisheries research. American Journal of Veterinary Research. 75: 402-416.
- Cooke, S.J., Wilson, A.D.M., Elvidge, C.K., Lennox, R.J., Jepsen, N., Colotelo, A.H. & Brown, R.S. (2015): Ten practical realities for Institutional Animal Care and Use Committees when evaluating protocols dealing with fish in the field. Reviews in Fish Biology and Fisheries.
-
Dance, M.A., Moulton, D.L., Furey, N.B. & Rooker, J.R. (2016): Does transmitter placement or species affect detection efficiency of tagged animals in biotelemetry research? Fisheries Research. 183: 80-85.
Further reading
- A collection of training templates (CCAC website)
- Approved protocols for decapods, cephalopods and fish (Guidance from the Animal Ethics Sub-Committee of the University of KwaZulu-Natal Ethics Committee, South Africa)
- Lambert H, Carder G & D'Cruze N (2019): Given the Cold Shoulder: A Review of the Scientific Literature for Evidence of Reptile Sentience
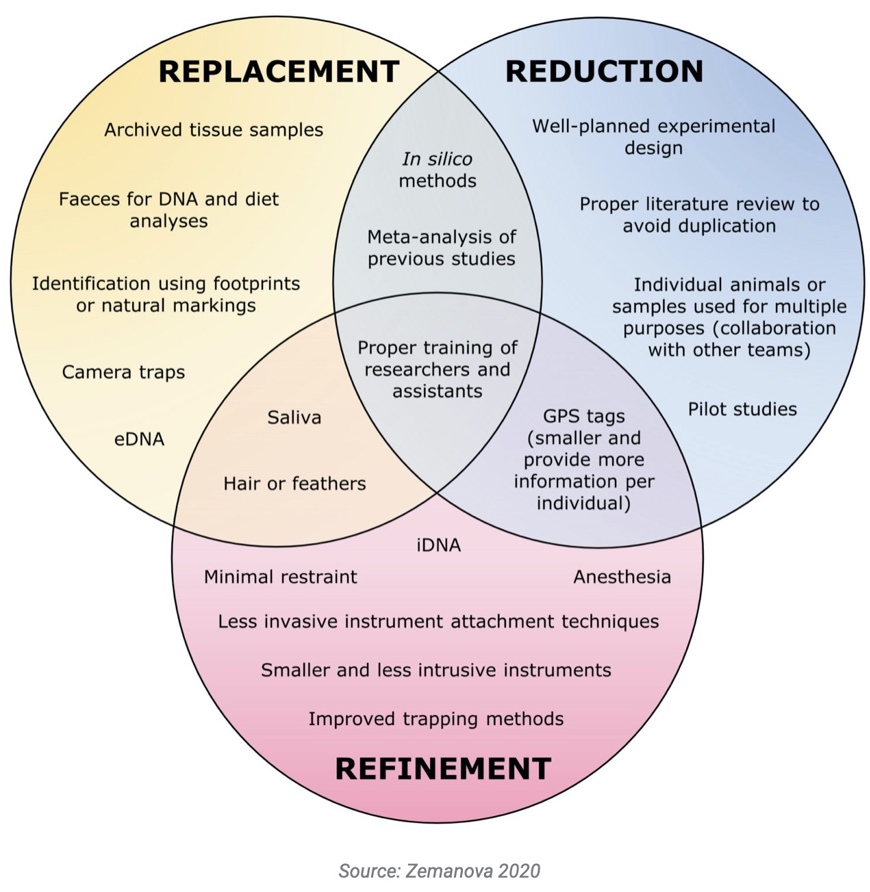
Image from Zemanova (2020)
An overview of animals used in research from Understanding Animal Research (UAR)
Thanks for your feedback! Please note that we cannot reply to you unless you send us an email.
What are you looking for?
We value your feedback so we can improve the information on the page. Please add your email address if you would like a reply. Thank you in advance for your help.!
Please contact us by email if you have any questions.

