Norwegian legislation on animal experimentation
Most of Norway's legislation is only available in Norwegian, but Norway's Animal Welfare Act is available in English.
Norwegian legislation concerning the use of animals in research and testing has been amended to implement the requirements contained in EUs Directive 2010/63 on the use of animals for scientific purposes. Norway fully implemented the Directive on 1 August 2016 via the European Economic Area Agreement (EØS-avtalen). This includes the establishment of a National Committee for the Protection of Animals used for Scientific Purposes.
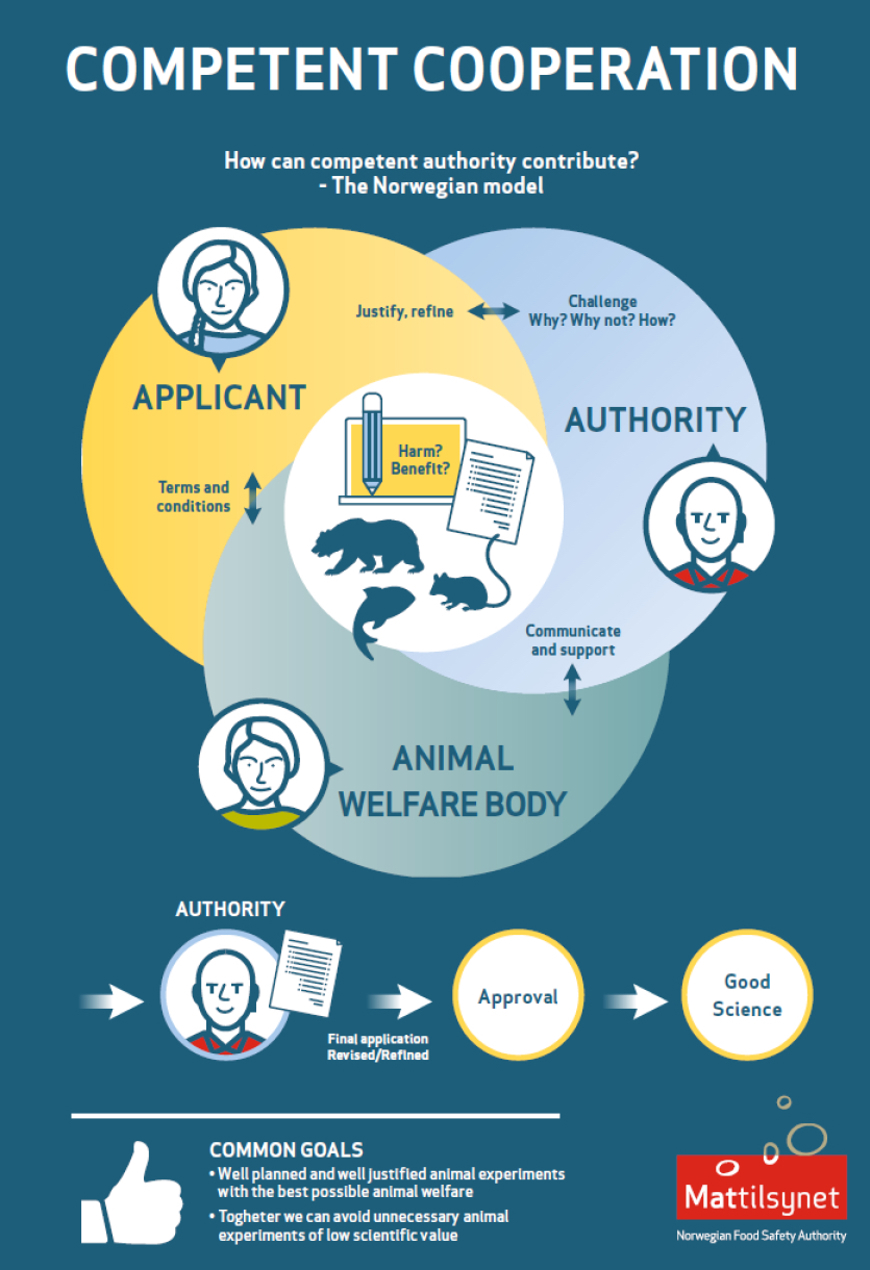
The National Assignments Department at the Norwegian Food Safety Authority handle all applications for animal research (experiments and user establishments), undertake retrospective assessment, issue reports, implement Directive 2010/63, conduct inspections, give advice and answer transparency enquiries. Staff members also function as Norway's PARERE contact, as the Secretary of Norway's National Committee for the Protection of Animals Used for Scientific Purposes, and as the National Contact Point for the EU Commission. They can be contacted at forsoksdyr@mattilsynet.no
The Authority's Head Office handles complaints and performs regulatory development.
Non-Technical Summaries (NTS) of approved projects (page in Norwegian)
The Authority undertakes harm-benefit assessments and ethical evaluation of applications for animal use. It also uses the ethical guidelines issued by the National Committee for Research Ethics in Science and Technology (NENT). This Committee has also produced general guidelines for research ethics in science and technology, which contain sections on the protection of animals used in research.
In the case of fish research and fish farming, the Regulation on aquaculture is also highly relevant, in particular paragraph 20 which states that methods, installations and equipment can only be used when their consequences for the animals' welfare has been documented. The Food Safety Authority decides whether this animal testing falls under the Regulation for the use of animals in experiments.
On the Norwegian version of this page, the use of genetically altered animals and micro-organisms is also discussed.
See also the report of the Norwegian Scientific Committee for Food and Environment (VKM, 2024) on marking of wild birds (in Norwegian).
Statistics on the use of animals in research, testing and education are available here.
For comparison
- Denmark
- The Animal Experiments Council (Rådet for Dyreforsøg)
- The Animal Experiments Inspectorate (Dyreforsøgstilsynet) (the secretariat for the Council)
- The Danish 3R-Center
- The National Committee for the Protection of Animals Used for Scientific Purposes (Udvalget for Forsøgsdyr og Alternativer) (Denmark's national committee; the committee members are also the Board of the 3R-Center)
Thanks for your feedback! Please note that we cannot reply to you unless you send us an email.
What are you looking for?
We value your feedback so we can improve the information on the page. Please add your email address if you would like a reply. Thank you in advance for your help.!
Please contact us by email if you have any questions.
