Relationship between PREPARE and ARRIVE
The PREPARE guidelines for planning animal experiments are complementary to the many reporting guidelines such as ARRIVE. Although important in itself, better reporting cannot improve the quality, reproducibility and translatability of animal studies. Improvements require better planning, from day one, in close collaboration with animal care staff. Both types of guideline should be used, to ensure that important topics are not omitted at each stage of the process from the conception of research ideas to publication. This will also improve the validity, reproducibility and translatability of animal studies, as well as identify important financial and health & safety issues. We have illustrated this in a whiteboard film.
We have made a collection of endorsements of PREPARE.
PREPARE includes several topics which may not be necessary to report in a scientific paper, but which can seriously affect the scientific outcome, costs, animal welfare and the health & safety of both humans and animals.
Here is a 4-step approach for scientists conducting animal studies:
- PREPARE: Use the PREPARE guidelines from day 1 of planning, to implement the 3Rs and identify all the factors which can affect the validity of their research
- CARE: Work to develop a Culture of Care at the research facility. This is a commitment to improving animal welfare, scientific quality, care of the staff and transparency for the stakeholders.
- SHARE: Use reporting guidelines to write a manuscript which adequately describes the study. Ensure that any refinements developed during the study are shared: for example in separate method papers, or in the Refinement Wiki.
- FLAG: Make sure that any advances in the 3Rs which were made during the study are prominently mentioned in the manuscript, preferably in the title and/or abstract, since many databases only index these parts of a paper.
We have made a webpage with freely downloadable icons to illustrate this.
Further reading
- Twelve years after the ARRIVE guidelines: Animal research has not yet arrived at high standards(Song et al., 2023)
- Animal welfare requirements in publishing guidelines (Novak et al., 2022)
- Improving Replicability, Reproducibility and Reliability In Preclinical Research: A Shared Responsibility (Cheleuitte-Nieves & Lipman, 2019)
- Reporting guideline checklists are not quality evaluation forms: they are guidance for writing (Logullo et al., 2020)
- Improving Reproducibility and Translatability in Shock: the Arrive Guidelines Need Better Implementation and Enforcement (Reynolds, 2020)
- The ARRIVE 2.0 Essential 10: Guidance for NIH-sponsored Research (webinar by Penny Reynolds, 15 June 2023)
An infographic from Utrecht University showing the path to better research:
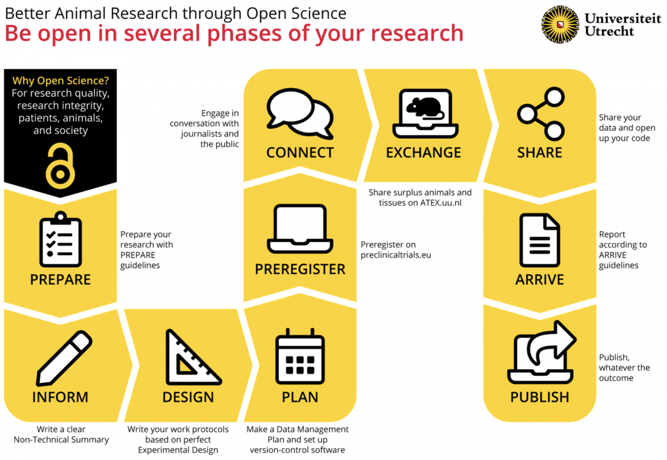
With permission from Utrecht University. Published in Janssens et al. (2023).

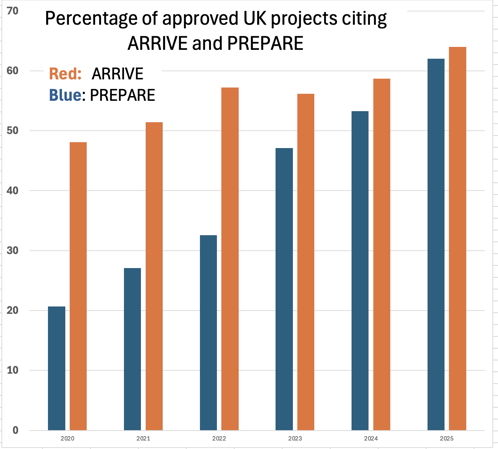
An analysis of the uptake of ARRIVE and PREPARE. The columns show the precentage of Non-Technical Summaries of projects approved by the UK Home Office from 1 January 2019 to 31 March 2025 which stated that they used these guidelines in connection with the projects.